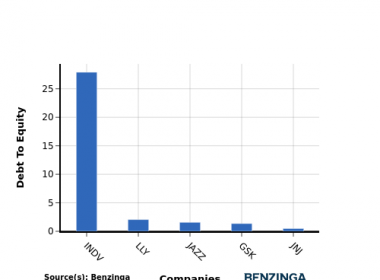
AbbVie’s Arthritis Drug Humira Retains Market Dominance Despite Biosimilar Competition, Challenges Biosimilar Industry Viability
Humira maintains over 80% of its market share despite facing nine biosimilar rivals, raising concerns about the future of the biosimilar market and prompting calls for regulatory reform to enhance patient access and incentivize development.