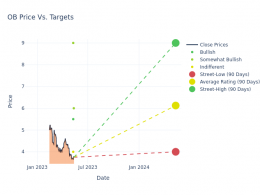
- Last month, Angion Biomedica Corp (NASDAQ:ANGN) discontinued the JUNIPER Phase 2 dose-finding trial of ANG-3070 in primary proteinuric kidney diseases, specifically focal segmental glomerulosclerosis and immunoglobulin A nephropathy.
- HC Wainwright downgraded the stock to Neutral from Buy without a price target.
- The analysts say that Angion continues to pursue a Phase 1b study of ANG-3070 in patients with idiopathic pulmonary fibrosis (IPF). Topline data remain scheduled to release later this year.
- “While we do not expect to get a sense of efficacy from this trial, it may pave the way for ANG-3070 to move into proof-of-concept IPF clinical assessment next year. We also expect safety and tolerability data in IPF patients to be generated from the Phase 1b trial,” notes HC Wainwright.
- “While we remain cautiously optimistic regarding ANG-3070’s potential in IPF, we recognize that this will take time to realize. In the wake of the JUNIPER discontinuation,” the analysts added.
- Price Action: ANGN shares are down 2.36% at $1.04 during the market session on the last check Monday.
India’s Reliance Maintains Silence On Mega Merger Buzz With Disney’s Local Unit
This story was first published on the Benzinga India portal.